Plus Two Physics Chapter 12 Atoms Chapter Wise Question and Answers PDF Download: Students of Standard 12 can now download Plus Two Physics Chapter 12 Atoms chapter wise question and answers pdf from the links provided below in this article. Plus Two Physics Chapter 12 Atoms Question and Answer pdf will help the students prepare thoroughly for the upcoming Plus Two Physics Chapter 12 Atoms exams.
Plus Two Physics Chapter 12 Atoms Chapter Wise Question and Answers
Plus Two Physics Chapter 12 Atoms question and answers consists of questions asked in the previous exams along with the solutions for each question. To help them get a grasp of chapters, frequent practice is vital. Practising these questions and answers regularly will help the reading and writing skills of students. Moreover, they will get an idea on how to answer the questions during examinations. So, let them solve Plus Two Physics Chapter 12 Atoms chapter wise questions and answers to help them secure good marks in class tests and exams.
|
Board |
Kerala Board |
|
Study Materials |
Chapter wise Question and Answers |
|
For Year |
2021 |
|
Class |
12 |
|
Subject |
Physics |
|
Chapters |
Physics Chapter 12 Atoms |
|
Format |
|
|
Provider |
How to check Plus Two Physics Chapter 12 Atoms Question and Answers?
- Visit our website - https://spandanamblog.com
- Click on the 'Plus Two Question and Answers'.
- Look for your 'Plus Two Physics Chapter 12 Atoms Question and Answers'.
- Now download or read the 'Class 12 Physics Chapter 12 Atoms Question and Answers'.
Plus Two Physics Chapter 12 Atoms Question and Answers PDF Download
We have provided below the question and answers of Plus Two Physics Chapter 12 Atoms Chapter wise study material which can be downloaded by you for free. These Plus Two Physics Chapter 12 Atoms Chapter Wise Question and answers will contain important questions and answers and have been designed based on the latest Plus Two Physics Chapter 12 Atoms, books and syllabus. You can click on the links below to download the Plus Two Physics Chapter 12 Atoms Chapter Wise Question and Answers PDF.
Question 1.
What is the shortest wavelength present in the Paschen series of spectral lines?
Answer:
Since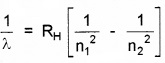
For Paschen series, n1 = 3 and n2 = ∞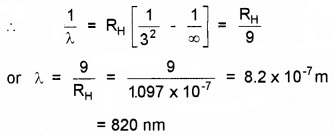
Question 2.
The radius of the innermost electron orbit of a hydrogen atom is 5.3 × 10-11 m. What are the radii of the n = 2 and m = 3 orbits?
Answer:
Given r1 = 5.3 × 10-11m
n1 = 1, n2 = 2, r2 = ?
n3 = 3, r3 = ?
Since r ∝ n2
∴ r1 ∝ n12
And r2 ∝ n22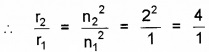
or r2 = 4 × r1 = 4 × 5.3 × 10-11
or e2 = 2.12 × 10-10m
Also r3 ∝ n32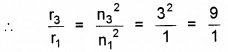
or r3 = 9r1 = 9 × 5.3 × 10-11
or r3 = 4.77 × 10-10m.
Question 3.
A 12.5 eV electron beam is used to bombard gaseous hydrogen at room temperature. What series of wavelength will be emitted?
Answer:
First excitation energy
E = E2 – E1 = -3.4 – (-13.6) = 10.2 eV
Second excitation energy
E = E3 – E1 = -1.51 -(-13.6) = 12.09 eV
Third excitation energy
E = E4 – E1 = -0.85 – (-13.6) = 12.75 eV
Second incident beam has energy = 12.5 eV
So only first two lines in the Lyman series of wave-length 103 nm and 122 nm will be emitted.
Also E3 – E2 = -1.51 – (-3.4) = 4.91 eV
i.e, first line in the Balmer series of wavelength 656nm will also be emitted.
Question 4.
In accordance with the Bohr’s model, find the quantum number that characterises the earth’s revolution around the sun in an orbit of radius 1.5 × 1011 m with orbital speed 3 × 104 ms-1.
Answer:
Here r= 1.5 × 1011 m,
v = 3 × 104 ms-1, m = 6 × 1024kg, n = ?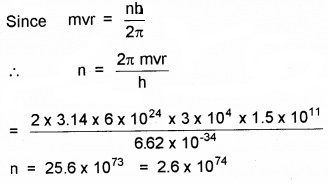
Plus Two Physics Atoms One Mark Questions and Answers
Question 1.
A radioactive nucleus emits beta particle. The parent and daughter nuclei are
(a) isotopes
(b) isotones
(c) isomers
(d) isobars
Answer:
(d) isobars
Question 2.
Consider an electron in the nth orbit of a hydrogen atom in the Bohr model. The circumference of the orbit can be expressed in terms of de Broglie wavelength λ of that electron as
(a) (0.529) nλ
(b) \(\sqrt{n} \lambda\)
(c) (13.6)λ
(d) nλ
Answer:
(d) nλ
Question 3.
The ionization energy of hydrogen atom is 13.6 eV. Find the energy corresponding to a transition between 3rd and 4th orbit.
Answer:
E = E4 – E3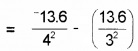
= -0.85 + 1.51 = 0.66 eV.
Question 4.
If 13.6 eV energy is required to ionize the hydrogen atom, then the energy required to remove an electron from n = 2 is
(a) 10.2 eV
(b) 0 eV
(c) 3.4 eV
(d) 6.8 eV
Answer:
(c) 3.4 eV
Explanation:![]()
= 3.4 eV.
Question 5.
In the Bohr model of the hydrogen atom, the lowest orbit corresponds to
(a) infinite energy
(b) maximum energy
(c) minimum energy
(d) zero energy
Answer:
(c) minimum energy.
Question 6.
Write down the Balmerformula for wavelength of Ha line.
Answer: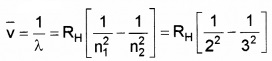
Plus Two Physics Atoms Two Mark Questions and Answers
Question 1.
Given Rydberg constant as 1.097 × 10-7m-1. Find the longest and shortest wavelength limit of Baler Series.
Answer:
\(\bar{v}=\frac{1}{\lambda}=R_{H}\left[\frac{1}{n_{1}^{2}}-\frac{1}{n_{2}^{2}}\right]\)
Longest wavelength n1 = 2 and n2 = 3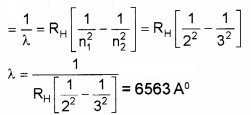
Shortest Wavelength n1 = 2 and n2 = α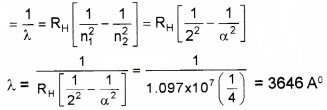
Plus Two Physics Atoms Three Mark Questions and Answers
Question 1.
Bohr combined classical and early quantum concept and gave his theory in the form of three postulates.
- The total energy of an electron in ground state of hydrogen atom is -13.6eV. What is the significance of negative sign?
- The radius of innermost electron orbit of hydrogen atom is 5.3 × 1011m. What are the radii of n = 2 and n = 3 orbits?
Answer:
1. Negative sign implies that the electrons are strongly bounded to the nucleus.
2. rn = n2a0 = 5.3 × 10-11m
r1 = a0 = 5.3 × 10-11m
r2 = 4a0 = 21.2 × 10-11m
r3 = 9a0 = 47.7 × 10-11m.
Plus Two Physics All Chapters Question and Answers
- Plus Two Physics Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 1 Electric Charges and Fields Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 2 Electric Potential and Capacitance Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 3 Current Electricity Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 4 Moving Charges and Magnetism Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 5 Magnetism and Matter Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 6 Electromagnetic Induction Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 7 Alternating Current, Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 8 Electromagnetic Waves Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 9 Ray Optics and Optical Instruments Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 10 Wave Optic Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 11 Dual Nature of Radiation and Matter Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 12 Atoms Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 13 Nuclei Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 14 Semiconductor Electronics: Materials, Devices and Simple Circuits Chapter Wise Question and Answers PDF
- Plus Two Physics Chapter 15 Communication Systems Chapter Wise Question and Answers PDF
Benefits of the Plus Two Physics Chapter 12 Atoms Chapter Wise Question and Answers PDF
The Plus Two Physics Chapter 12 Atoms Chapter Wise Question and Answers PDF that has been provided above is extremely helpful for all students because of the way it has been drafted. It is designed by teachers who have over 10 years of experience in the field of education. These teachers use the help of all the past years’ question papers to create the perfect Plus Two Physics Chapter 12 Atoms Chapter Wise Question and Answers PDF.
0 comments:
Post a Comment